Advancing the use of computational modeling to support alternative bioequivalence approaches in alignment with expectations of major regulators.
Nanopharm and FLUIDDA’s SmartTrack™ is an integrated digital development platform for inhaled drug products that utilizes real world patient data combined with advanced in vitro models and in silico simulations to help demonstrate bioequivalence for generic or reformulated products.
The use of the SmartTrack™ computational modelling approach can help to derisk inhaled drug development and reduce the risk of potential regulatory deficiencies.
Bioequivalence (BE) is defined as the demonstration that two drug products release their active ingredients at the site of action at the same rate and extent when taken under the same conditions.
According to the U.S. FDA’s traditional “Weight of Evidence” approach, in order to demonstrate bioequivalence (BE), the developer must be able to demonstrate that the formulation and device are similar, in vitro tests are equivalent, systemic pharmacokinetic (PK) exposure is equivalent and pharmacodynamics (PD) or comparative clinical endpoint (CCEP) efficacy are equivalent.
Since 2024, the need for a CCEP study has been no longer mandatory in lieu of additional in vitro and PK studies. This change appears to reduce the burden on the developer, but in practice has led to other challenges which have arisen following the implementation of the additional tests.
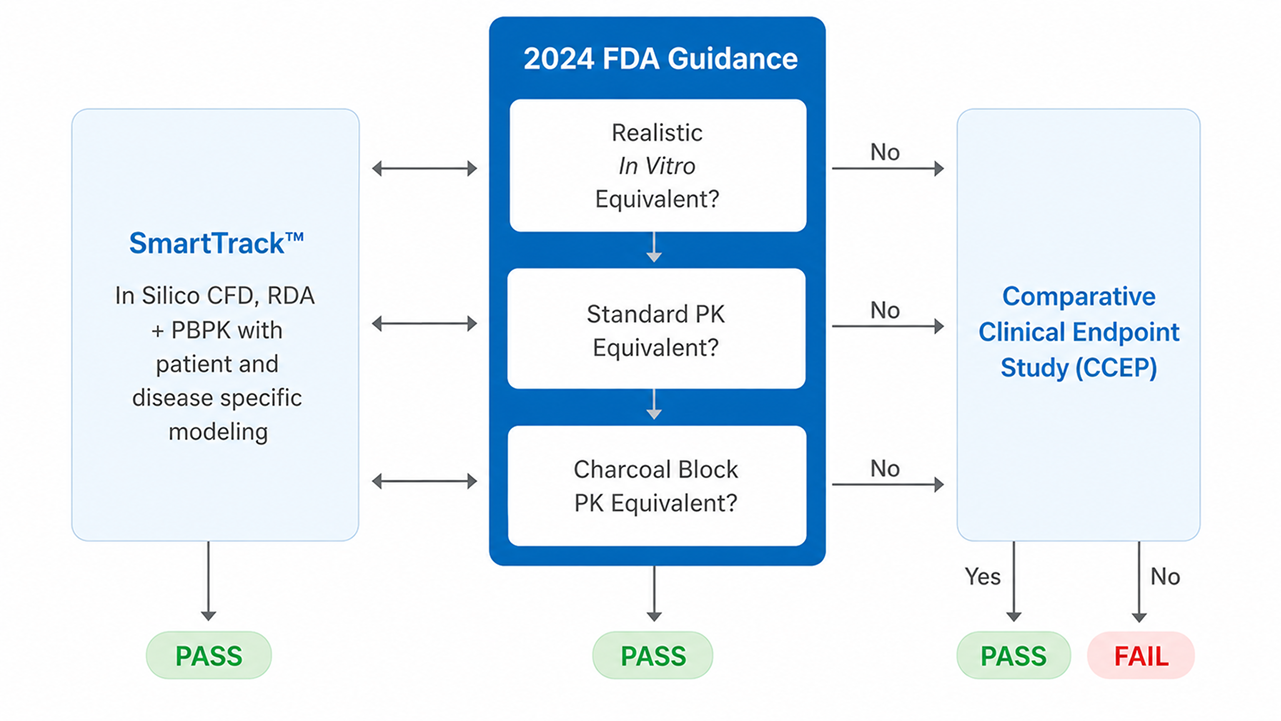
If any of the mandatory elements were to fail, SmartTrack™ can be applied to demonstrate whether or not these differences would really impact the comparative performance in a clinical setting and potentially support a justification of bioequivalence without going back to CCEP studies. Without computational modelling approaches, there is no alternative mitigation if there are any failures in the in vitro or clinical segments. Doing this proactively, rather than reactively, will derisk and accelerate the inhaled drug product development program.
Nanopharm has initiated a series of clinical studies to measure PK and regional lung deposition (through gamma scintigraphy) on a number of marketed pMDI drug products. The clinical results will be compared to simulations made using SmartTrack™’s modeling tools to validate the platform, ultimately for inclusion in a Model Master File (MMF).
The study has been designed based on feedback from the U.S. FDA and will cover an operating space of existing pMDI products, so that sponsors would often not have to repeat the validation activity for their own dossiers. The modeling technology will ultimately be validated according to the U.S. FDA Guidance for “Assessing the Credibility of Computational Modelling and Simulation”* The program is expected to be completed by late 2026.
* (Assessing the Credibility of Computational Modeling and Simulation in Medical Device Submissions | FDA), based on the ASME V&V40 framework, with different risk profiles and use cases incorporated.
Nanopharm is generating a Model Master File (MMF) for SmartTrack™, based on similar principles to a Drug Master File (DMF). An MMF is a confidential, regulator reviewed summary of the SmartTrack™ platform’s model covering the structure, assumptions, validation data and performance of the computational model.
Sponsor companies will be able to then reference the MMF in their drug product filings without the need to reperform the same activities – saving time, cost and risk to their bioequivalence program for inhaled drug products.
The U.S. FDA is already referring to the use of computational or in silico modelling, like SmartTrack™, as an optional approach to demonstrating bioequivalence in a number of recent Product Specific Guidances (PSGs) implying that this modelling is recommended rather than the other way around.
Other administration routes are already far more established in this regard, and the complexity of inhaled drug products is making it harder to find a viable solution. The update to the PSGs is a significant sign of intent from the agency on their willingness to adopt these approaches as supportive evidence.


Nanopharm has engaged with the U.S. FDA through sponsored workshops, research grants and broader collaborations for over 10 years to help better understand and define alternatives approaches to CCEP studies s for inhaled drug products.
along with Nanopharm and FLUIDDA to build its knowledge around modelling for OINDPs. These studies include , including for a next generation propellant (NGP) pressurized metered dose inhaler (pMDI) study.
have been held with Pharma Sponsors to move towards alignment on alternative approaches to bioequivalence, including for lower Global Warming Potential (GWP) propellant driven pMDIs.