SmartTrack™ is an alternative digital platform using real patient data with in vitro and in silico models to support bioequivalence.
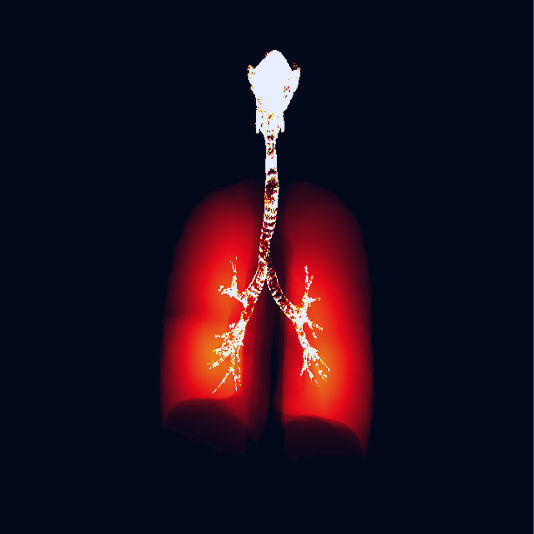
SmartTrack™ is an advanced alternative bioequivalence platform that accelerates and derisks inhaled drug product development through an integrated digital platform that utilizes real world patient data combined with advanced in vitro tests and in silico simulations to support claims of bioequivalence for generic orally inhaled and nasal drug products (OINDPs). The SmartTrack™ platform has been developed over the last decade to support the justification of biowaivers for comparative clinical endpoint (CCEP) studies with ongoing interaction with the U.S. FDA.
SmartTrack™ can provide sponsors with shorter inhaled drug development times along with data driven support for regulatory filings.

Real-world patient breathing profiles

Method development using CFD
PK simulation via PBPK

Realistic in vitro aerosol testing and lung deposition modelling using CFD and RDA

*Validation study ongoing – expected completion end 2026.
SmartTrack™ is a faster and more data-driven alternative pathway to build a solid base of evidence and understand the root cause of the observed differences between clinical and in vitro datasets.
SmartTrack™ can help to reduce the ambiguity and number of follow-on questions from regulators by providing a comprehensive weight of evidence analysis of the bioequivalence data.
The SmartTrack™ platform answers many of the common questions sponsors/drug developers have about how they derisk and accelerate the development of their inhaled drug products
Not only is SmartTrack™ an ideal digital bioequivalence platform for generic orally inhaled drug products, but it also offers significant benefits to many other OINDP applications.

Nanopharm and FLUIDDA's SmartTrack™ is an integrated digital evidence platform that can demonstrate bioequivalence faster and reduce reliance on comparative clinical endpoint (CCEP) studies. It does so in a single integrated simulation framework that reliably predicts the real-world performance of inhaled drug products in humans.