Generic Inhaled Drug Product Development
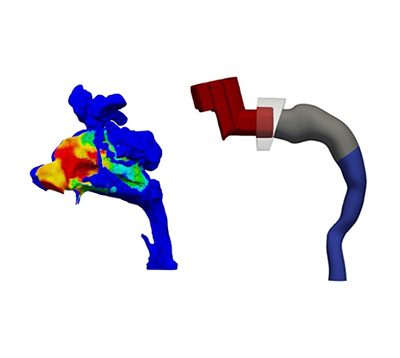
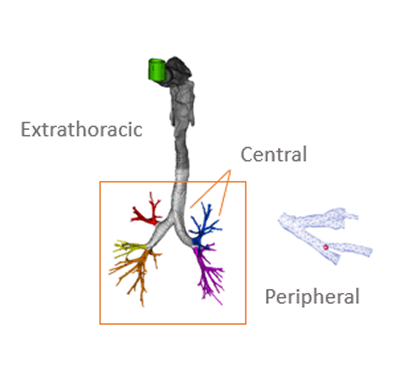
SmartTrack™ can be used to help demonstrate the bioequivalence of a new generic inhaler drug product to a reference listed drug (RLD), supporting justifications around potentially overdiscriminating in vitro data, without having to default to costly, time consuming comparative clinical endpoint studies (CCEPs) that lack sensitivity.