FLUIDDA's FRI integrates HRCT and CFD to characterize individual lung geometries and quantifies the impact of disease on lung function and inhaled aerosol drug deposition.
Through an exclusive partnership between Nanopharm and FLUIDDA, who is recognized as a global leader in Functional Respiratory Imaging (FRI), SmartTrack™ includes a unique, patient-specific lung database that underpins its inhaled drug development platform.
FRI integrates High-Resolution Computed Tomography (HRCT) with advanced Computational Fluid Dynamics (CFD) to characterize individual lung geometries and quantify the impact of disease on lung function and inhaled aerosol deposition.
Selection and optimization of inhalation delivery systems
Guides formulation strategies to achieve robust demonstrations of BE or lung dose optimization
Takes a fraction of the computational time and cost
Significantly improves scalability and turnaround in drug development workflows
In contrast, conventional lung function metrics such as FEV₁ or FVC provide only global assessments of lung function and lack the regional resolution needed to understand airflow distribution and drug deposition at a local level, making them limited in value for proactively accelerating and derisking inhaled drug development while struggling to provide meaningful and credible data to regulators.
 1
1
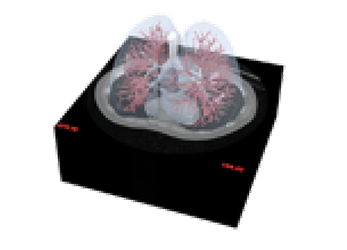
HRCT scans use X-rays to create a highly detailed, thin-slice image of the lungs
 2
2
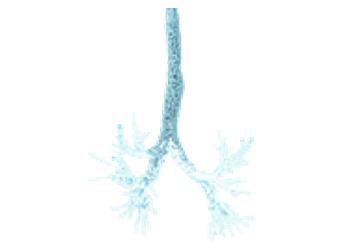
Segmented 3-dimensional geometries are measured from the HCRT scans of patient lungs
 3
3
Computational Fluid Dynamics (CFD) and proprietary Rapid Deposition Analysis (RDA) quantifies airflow and exposure to inhaled particles regionally in the lungs using 3D computational models for a representative flow simulation
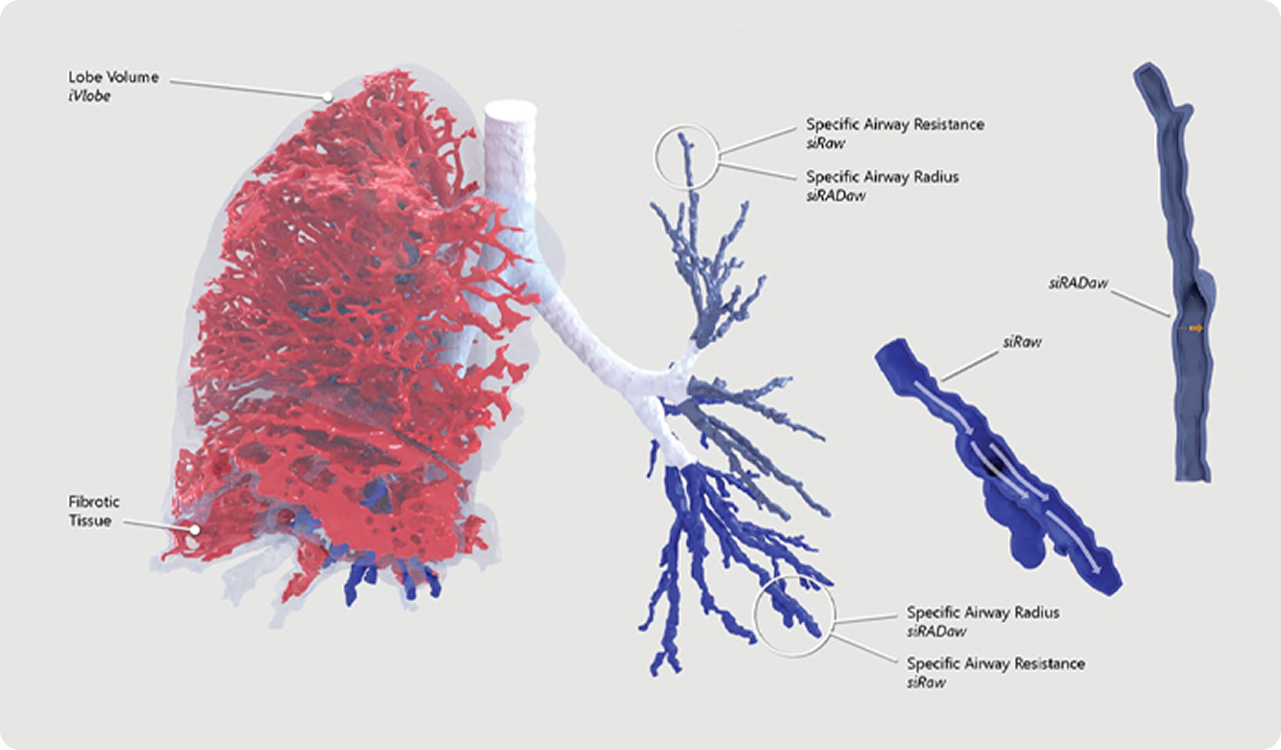
The result of FRI is a clinically meaningful measurement of patient-specific respiratory systems that define a set of biomarkers used to analyze the exposure to inhaled drugs as influenced by the structure and function of the lungs and airways. Disease-specific biomarker models can be developed for specific lung diseases providing a regional understanding of lung pathophysiology (changes to the lungs of patients), that could impact the quantity and availability of inhaled drug reaching the desired location. FRI helps to quantify deposition regionally and visualize the lungs realistically.

Functional Respiratory Imaging (FRI) is capable of capturing virtually all key sources of variability that drive regional lung exposure following the inhalation of medication; factors that are also encountered in clinical trials of inhaled therapies. This variability is a primary driver of clinical trial design, directly influencing sample size, study duration, and the associated costs which are often substantial.
These sources of variability include inhaler geometry, inhalation profiles, formulation characteristics, as well as patient and disease specific lung structure and function. By explicitly accounting for these parameters, FRI enables a highly granular and mechanistic understanding of drug deposition and effect.
As a result, FRI makes SmartTrack™ a uniquely comprehensive platform in the market, enabling stakeholders such as drug developers and regulators to gain detailed insight into the performance of inhaled drugs without having to rely on large, costly clinical trials. This proprietary capability has the potential to accelerate the development of novel therapies, as well as streamline the approval of generics and metered dose inhalers using next generation propellants.

FLUIDDA’s approach to FRI in collaboration with Nanopharm provides the SmartTrack™ platform with a unique insight into lung airflow, which can impact the delivery of inhaled drug products, based on Computational Fluid Dynamics (CFD) modeling built upon regionally specific lung geometries and data built from actual human subject data.

This unique FRI technology is a critical part of what makes SmartTrack™ an efficient, de-risking tool in the demonstration of bioequivalence of generic inhaled drug products.