SmartTrack™ is an alternative bioequivalence platform for inhaled drugs that combine in vitro and in silico models with real patient data.
Nanopharm and FLUIDDA’s SmartTrack™ alternative bioequivalence platform was designed through collaboration to accelerate inhaled drug product development through an integrated digital platform that utilizes real world patient data combined with advanced in vitro models and in silico simulations to support claims of bioequivalence for generic inhalers.

Patient breathing
profile collection

Microstructural characterization

In vitro realistic
aerosol testing

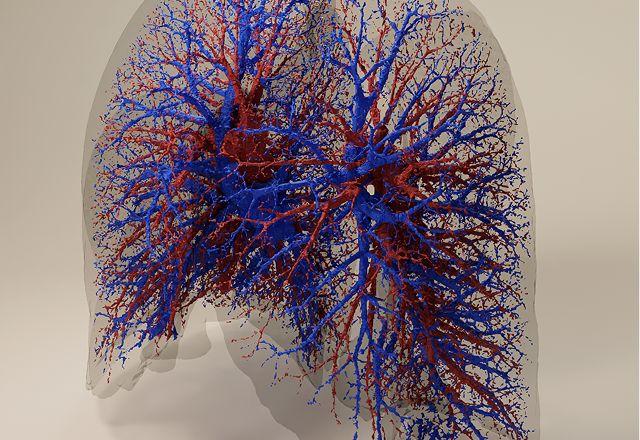
in silico regional lung deposition modeling in patient models

in silico
PK modeling
SmartTrack™ applies real patient breathing profiles and anatomical throat models in its in vitro testing and then models deposition in real patient airways using its in silico models. Together this produces a detailed and predictive understanding of how the patient, drug and device together impact drug delivery and help form a more holistic understanding of their bioequivalence to comparator products.

for bioequivalence comparisons using realistic aerodynamic particle size distribution (ASPD) and plume geometry studies, in case traditional limits for QC-based tests are overdiscriminating or misleading.
including human factors, such as the coordination between patient inhalation and actuation, on regional lung deposition using computational fluid dynamics (CFD), which may also impact in vitro data.
Validation study ongoing – expected completion end 2026.
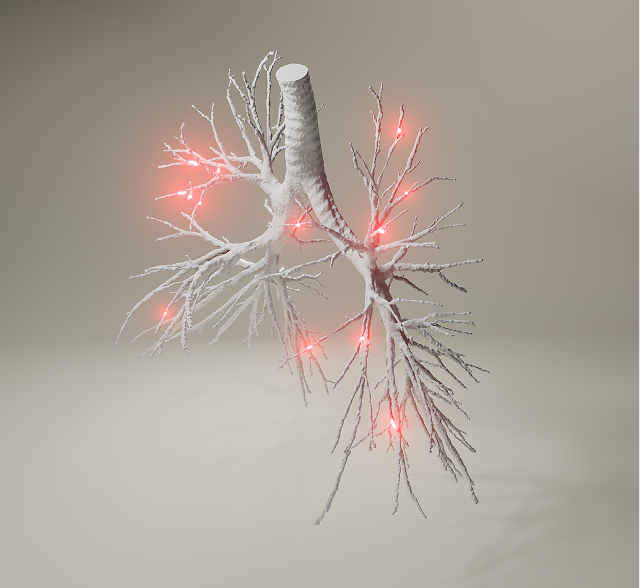
Traditional bioequivalence studies can lack the necessary sensitivity to fully account for human variability and the complexity of the inhaled drug delivery route on bioequivalence. Nanopharm and FLUIDDA’s SmartTrack™ platform connects experimental data and mechanistic simulations to create a single model that predicts end-to-end inhaled drug delivery performance.
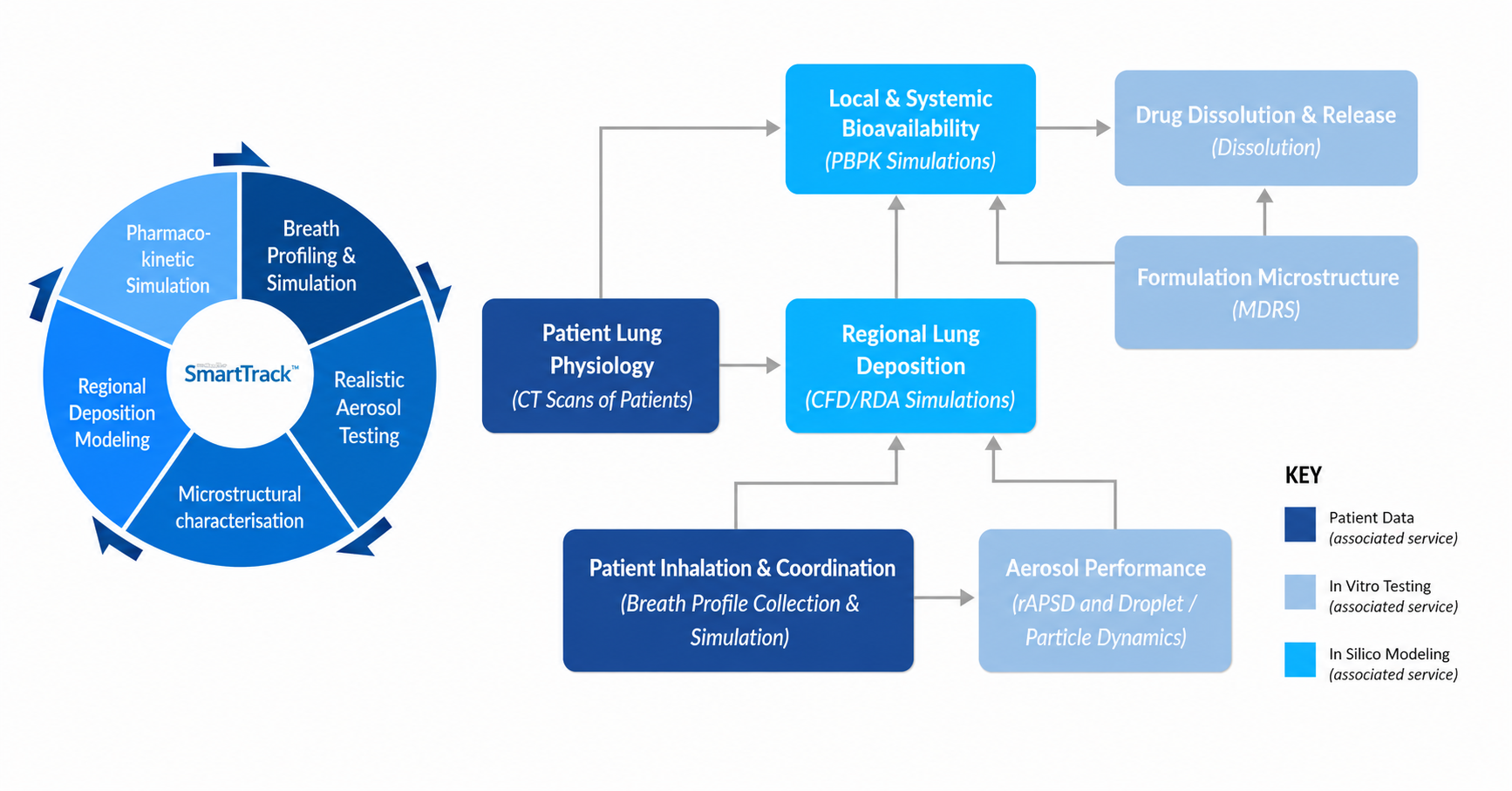
The SmartTrack™ alternative bioequivalence platform consolidates many of its specialized tests and proprietary models to deliver a more detailed and representative data-driven understanding of the actual presence and deposition of inhaled drugs in the lungs.
SmartTrack™ is based upon a multi-layered framework that integrates aspects of aerosol science, lung physiology, formulation microstructure, and computational modeling into a single, predictive system.
Since 2002, Nanopharm has been a leading specialist CRO for nasal and inhaled drug products offering formulation, analytical testing and computational pharmaceutics services. In collaboration with FLUIDDA, a world leader in the field of Functional Respiratory Imaging (FRI), the SmartTrack™ platform uses real patient inhalation data and lung geometries, integrated into purpose-built models, to separate product versus patient effects. Together, SmartTrack™ employs a number of specialized scientific approaches.

The SmartTrack™ platform integrates these foundational scientific elements together to produce a quantitative model that is able to reliably predict real world bioequivalence for inhaled drug products.
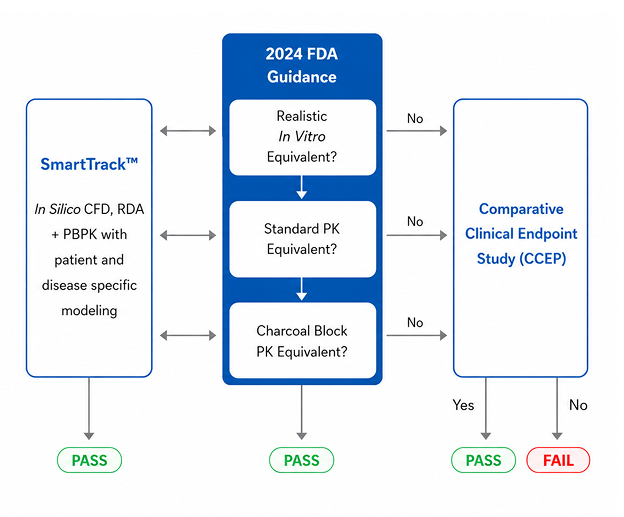
Nanopharm and FLUIDDA’s SmartTrack™ builds on the scientific foundations of inhaled drug delivery creating an integrated digital evidence platform that demonstrates bioequivalence. It does so in a single integrated simulation framework that reliably predicts the real-world performance of inhaled drug products in humans. It helps scientists and regulators to understand the variances evident in in vitro data and accounts for patient variability that may not be apparent in traditional systemic PK driven studies performed under a weight of evidence approach.
The SmartTrack™ alternative bioequivalence platform can reduce the need for comparative clinical endpoint (CCEP) studies in generic drug product approvals by accurately predicting clinical outcomes if in vitro data is inconclusive. This removes a major barrier for generic companies as well as regulators, thus paving the way for wider and earlier patient access to medications.