PBPK Modeling
A Physiologically-Based Pharmacokinetic (PBPK) model represents a mathematical description of a drug’s transportation through the body, from administration to excretion. PBPK models are composed of multi-layered differential equations that describe the physiological features associated with the ADME (absorption, distribution, metabolism, and excretion) processes that can be used in Pharmacokinetic (PK) investigations. Nanopharm has developed sophisticated proprietary PBPK models that can reliably be used to predict clinical PK during OINDP drug product development. This allows our scientists to develop an understanding of the drug’s rate/extent of release and bioavailability at the site of action before going into the clinic, and derisking your PK studies. Nanopharm’s PBPK models help to streamline the nasal or inhaled drug product development process.
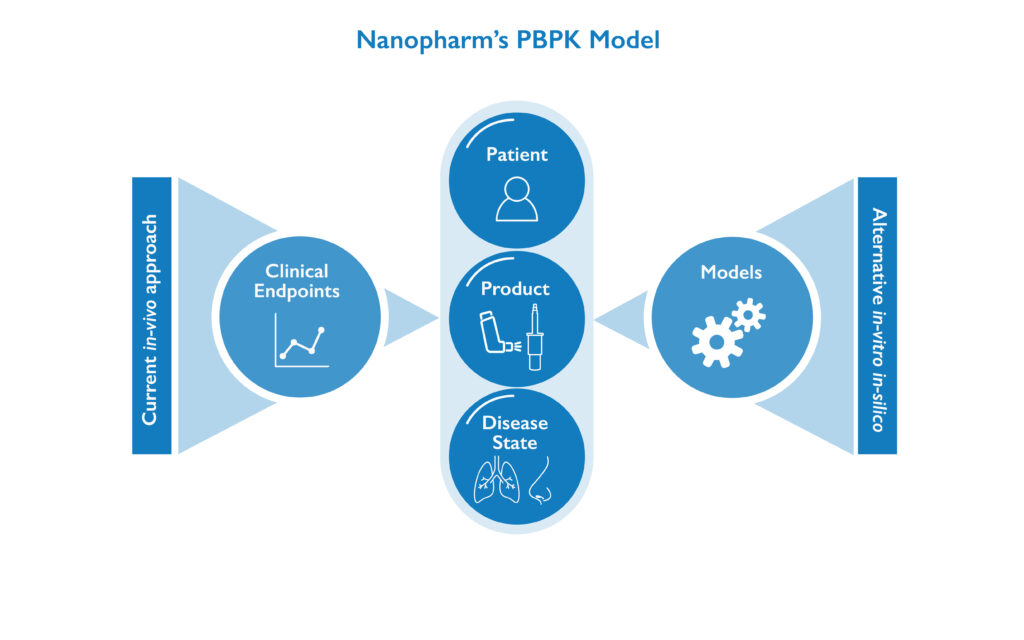
Nanopharm’s PBPK Modeling for OINDPs
Nanopharm’s PBPK modeling platform was developed to simulate the delivery of OINDP drug products, and specifically how they become available in the lungs or nose, since these are difficult to directly measure in the clinic. Our PBPK models can help us to predict how the drug products are absorbed into the bloodstream, as well as overall PK outcomes. Our PBPK models allow our scientists to effectively screen and optimize dosing regimens, excipients or enhancers as well as support device selection. The result is that Nanopharm can reliably predict the pharmacokinetics of inhaled and nasal drug products which can accelerate and derisk your OINDP product development.
Nanopharm’s OINDP PBPK models can accelerate drug product development and get you to the clinic faster through an in-silico simulation of the entire drug transportation process.
Nanopharm’s OINDP full body, bottom up PBPK platform is composed of models of individual transport processes. Our PBPK models can be built for most dosage forms, patient populations, or even a range of species.
OINDPs often act locally, at least in part, and local bioavailability is practically impossible to measure in the clinic. PBPK models predict duration of action in the lung or nose before the drug clears into the bloodstream.
Nanopharm’s PBPK Model Benefits for OINDP Development
Having access to robust PBPK modeling can help to accelerate OINDP development, thus moving into clinical trials faster and reducing costly rework throughout the entire drug development process. Drug transportation to the site of action for inhaled or nasal drug products can be particularly complex and difficult to understand. Patient physiology, technique, and endpoint sensitivity can make the identification of reliable biomarkers even more challenging because you cannot actually measure the local pharmacokinetics of OINDPs in the clinic. Nanopharm’s approach to PBPK modeling can identify the factors with the most impact on clinical efficacy, aid in investigating dosing regimens through techniques like drug dissolution and easily compare formulations / test products. These can help our respiratory scientists to select the optimal one as well as direct which parameters to manipulate in order to achieve the target PK profiles faster.
The OINDP format can have a major impact on pharmacokinetic (PK) profiles Therefore they must be accurately reflected in the PBPK in-silico models if they are to be representative and predictive in the patient. PBPK models can be adapted for every entry point into the body and follow drug transportation all the way through to the required point of action. For repurposed drugs, Nanopharm can first build a model for a marketed drug’s PBPK based on available data. It can then add on the inhaled or nasal drug entry pathway, incorporating the in-vitro and in-silico data, such as that created via our Aeronose™ nasal cast platform and our regional deposition models. Because our PBPK platform is built by modeling individual pharmacokinetic processes, the platform can be applicable to virtually all dosage forms, and a wide range of patient populations, or species. This allows us to take early-stage animal models and extrapolate to human models or new dosage forms.
As PBPK models capture drug transport from the entry point in the body to the site of action and ultimately their clearance pathways, the models must also describe how the drug is transported from the device into the body. PBPK models for inhalers require information on the aerosolization and deposition of the dose as it travels from the device into the nose or lung mucosa, through the epithelial layer and ultimately into the bloodstream. Nanopharm’s scientists can use computational fluid dynamics (CFD) together with the PBPK model to account for the complexity and interconnectivity of the biological structures involved. Nanopharm’s PBPK models incorporate regional deposition data with other in-vitro data such as cell permeability, mucoadhesion and dissolution to cover physiological factors that can impact bioavailability. Separate equations are used to describe the lung, the epithelial lung lining cells and the lung lining fluid. Nasal delivery is also complicated and requires the use of similarly complex models, with the factors such as mucociliary clearance to consider. For both inhaled and nasal drug delivery, it is always difficult to measure local tissue concentrations in order to validate results. Nanopharm’s scientists must balance the complexity of the model with what measurable and collectable data is available to populate the model, and still produce a useful predictive result. The PBPK models are built to meet the intended purpose, whether it’s to accelerate early OINDP development or if it must meet the highest standards required to support a bioequivalence (BE) claim for a generic product’s regulatory submissions.
Nanopharm PBPK Model Specialists for OINDPs
It takes dedication and a specialized focus on inhaled and nasal drug product delivery to build robust Physiologically-Based Pharmacokinetic (PBPK) models that have the ability to accelerate and derisk OINDP development or even support generic product bioequivalence claims with regulators. Nanopharm has the proven expertise in building reliable and predictive PBPK models for OINDPs that can move your inhaled or nasal drug product forward in the drug development process with data driven confidence.